HCQ COVID Studies-No Explanation for COVID Mortality Despite HCQ Treatments
Some studies have been retracted, some halted. The Henry Ford Study is the standout positive result. There is no randomized trial showing HCQ as effective for COVID. Not one. There are several against.
If HCQ was even as effective as TAMIFLU the results would have jumped right out as the academic equivalent of Captain obvious moments. TAMIFLU for the record id effective about 40% of the time against the flu. At this point, we have to ask the unasked question.
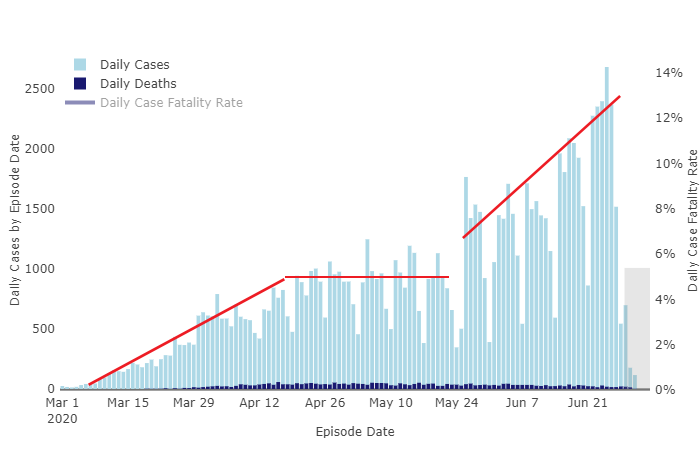
If HCQ is so damn good, why are people dying on it at a rate so close to the same as ordinary care we can’t pull the numbers out? Why is the support for it largely political and the science and medical fields largely against?
This resembles Climate Change. The argument against HCQ largely scientific. For? Political. When tobacco was still controversial as a cancer-causing habit, the opposition to the deal was commercial and political. Medical science won out in the end.
It will this time too. As of July 2020 there is no COVID cure. None. there is not yet a vaccine out of trials. What there is, is a resurgent virus and a Presidential election, a possible re election of the most visible champion a drug could have. The President of The United States. A politician and TV personality far more than a science guy.
Coincidence? I say not.
Off The top we have the Ford study, an example of a positive result for HCQ. But lets look at the details.
In a large-scale retrospective analysis of 2,541 patients hospitalized between March 10 and May 2, 2020 across the system’s six hospitals, the study found 13% of those treated with hydroxychloroquine alone died compared to 26.4% not treated with hydroxychloroquine.
A retrospective analysis. In other words, this is a look at data from an outside source, not a funded, designed, conducted an actual hands-on study. In common terms, it’s a hindsight data review.
And the author offers a cautionary note to the results and the kind of study that is far better at this kind of research.
Dr. Zervos also pointed out, as does the paper, that the study results should be interpreted with some caution, should not be applied to patients treated outside of hospital settings and require further confirmation in prospective, randomized controlled trials that rigorously evaluate the safety and efficacy of hydroxychloroquine therapy for COVID-19.
(my added bold)
The negative studies.
NEJM
A Randomized Trial of Hydroxychloroquine as Postexposure Prophylaxis for Covid-19
RESULTS
We enrolled 821 asymptomatic participants. Overall, 87.6% of the participants (719 of 821) reported a high-risk exposure to a confirmed Covid-19 contact. The incidence of new illness compatible with Covid-19 did not differ significantly between participants receiving hydroxychloroquine (49 of 414 [11.8%]) and those receiving placebo (58 of 407 [14.3%]); the absolute difference was −2.4 percentage points (95% confidence interval, −7.0 to 2.2; P=0.35). Side effects were more common with hydroxychloroquine than with placebo (40.1% vs. 16.8%), but no serious adverse reactions were reported.CONCLUSIONS
After high-risk or moderate-risk exposure to Covid-19, hydroxychloroquine did not prevent illness compatible with Covid-19 or confirmed infection when used as postexposure prophylaxis within 4 days after exposure. (Funded by David Baszucki and Jan Ellison Baszucki and others; clinicaltrials.gov number, NCT04308668. opens in new tab.)
Data from Solidarity (including the French Discovery trial data) and the recently announced results from the UK’s Recovery trial both showed that hydroxychloroquine does not result in the reduction of mortality of hospitalized COVID-19 patients when compared with standard of care.
A clinical trial to evaluate the safety and effectiveness of hydroxychloroquine for the treatment of adults hospitalized with coronavirus disease 2019 (COVID-19) has been stopped by the National Institutes of Health. A data and safety monitoring board (DSMB) met late Friday and determined that while there was no harm, the study drug was very unlikely to be beneficial to hospitalized patients with COVID-19. After its fourth interim analysis the DSMB, which regularly monitors the trial, recommended to the National Heart, Lung, and Blood Institute (NHLBI), part of NIH, to stop the study. NHLBI halted the trial immediately.
The Outcomes Related to COVID-19 treated with hydroxychloroquine among In-patients with symptomatic Disease study, or ORCHID Study, was being conducted by the Prevention and Early Treatment of Acute Lung Injury (PETAL) Clinical Trials Network of NHLBI. The data from this study indicate that this drug provided no additional benefit compared to placebo control for the treatment of COVID-19 in hospitalized patients.